Archiv
Dog owners watch out! Distemper virus in wild animals on the rise - more and more cases in Germany

Distemper cases are rampant this year in Germany among wild animals. Only a few days ago, cases were again reported in the Frankfurt city forest and in the Rhine-Siegkreis in North
Rhine-Westphalia. But also in various regions in Lower Saxony, Bavaria, Baden-Wuerttemberg and Rhineland-Palatinate deaths of wild animals by distemper accumulate. Besides martens and raccoons,
foxes in particular are infected with distemper. The positive rate here is sometimes over 80%.
Canine distemper virus (CDV), colloquially known as canine distemper, is a highly contagious viral infectious disease in dogs and carnivores that is often fatal. Lung and intestinal inflammation,
inflammation of the eye with accompanying blindness, as well as brain and nerve inflammation can occur in varying degrees during the course of the disease.
Since infection occurs through oronasal secretions (droplet infection), animals can become infected with canine distemper virus not only through direct contact with each other, but also through
excretions of any kind from infected animals, but also through contaminated water or objects. For unvaccinated dogs or dogs with insufficient vaccination protection, the risk of distemper
infection is therefore particularly high. To date, there is no antiviral therapy, symptoms caused by the disease can only be alleviated, but not cured.
Therefore, dogs and wild animals with distemper symptoms or suspicion should be treated immediately. Antigen detection in the form of rapid tests, which can be carried out immediately and without
technical effort in veterinary practices and provide a result within a few minutes, provide rapid certainty.
Due to the acute danger for dogs and wild animals caused by more and more distemper cases in our region, caution is required. With the help of antigen tests, current distemper cases can be
detected. With a proven vaccination protection we can go with our dogs without worries also in possible risk areas. The distemper vaccination belongs to the core vaccines. These vaccinations are
classified as necessary by the Ständige Impfkommission Veterinärmedizin (StIKo) in Germany, so that as many animals as possible can be protected. After basic immunization, it should be checked
after the third year whether there is sufficient immune protection or whether a booster vaccination is necessary. For this reason, high-ranking experts worldwide, such as the World Small Animal
Veterinary Association (WSAVA) and the Standing Commission on Vaccination in Veterinary Medicine (StIKo), recommend that, if possible, every animal should carry protective vaccine antibodies
against the canine distemper virus.
Especially in the danger areas, dog owners should be attentive, check the vaccination cards and, if necessary, have the dog's current immune protection checked with an antibody test at their
veterinarian. Here, too, there are already rapid tests for checking antibody titers. These are very inexpensive and show the vaccination status of the animal within a few minutes. Thus, not only
the antigen detection is an important tool to correctly assess the danger districts, but also the antibody detection is an important diagnostic tool to determine the immune status of the dogs, so
that a risk of infection is averted. Support and assistance with regard to diagnostics is offered by the German developer and manufacturer of rapid tests in the veterinary field, Fassisi GmbH
from Göttingen. The antigen detection of distemper (Fassisi CanDis) and a vaccine titer control for the viral diseases distemper, parvo
and adeno (Fassisi CantiCheck Plus) are also available here.
Modern vaccination management in dogs in practice
(20.09.2022) "Vaccinate as many dogs as possible, but only as often as necessary" is the motto of experts worldwide, such as the World Small Animal Veterinary Association (WSAVA) or the
Standing Commission on Vaccination in Veterinary Medicine (StIKo Vet).
With this demand, the current vaccination management in the veterinary sector must be generally reconsidered and modernised. But how can this demand of the experts be implemented in practice and
how can a modern, holistic vaccination management be established for the dog?
Vaccination management in dogs in practice Discussions about the frequency of vaccinations are becoming increasingly vocal in society. There is a broad consensus that the necessity of the
recommended vaccinations is generally undisputed and necessary. As many animals as possible should be given the core vaccines and build up sufficient immunity.
According to the WSAVA, the core vaccines should not be administered more frequently than every three years after the first vaccination series. Thus, a booster vaccination should be given after
three years at the earliest. However, this only applies to animals that no longer have any immune protection and for which a booster vaccination is therefore actually necessary.
The reason why not all animals have to be revaccinated across the board after 3 years is simple: the duration of immunity (DOI) is many years and can last until the end of the animal's life.
Above all, according to the WSAVA, it is completely pointless to vaccinate animals more frequently than necessary in order to increase the antibody titre.
In the case of existing immune protection, too early booster vaccinations do not increase the antibody titre level and thus do not generate additional immune protection.
On this basis, modern vaccination management dictates that each individual animal should be vaccinated as infrequently as necessary. But how can this be implemented in practice?
First, it must be determined individually whether the individual animal has sufficient immune protection. Current guidelines (for example, from the WSAVA) confirm that the presence of antibodies
against parvo, distemper and adenovirus, regardless of the titre level, indicates protective immunity and immunological memory.
For these 3 parameters, the demand of the Vaccination Commission for individual vaccination management in dogs can thus be implemented in practice by determining the antibodies present.
For the so-called antibody titer test, the individual antibodies of the dog are determined in a blood sample. In the opinion of the WSAVA, vaccination against parvo, distemper and adenovirus
should only be carried out if the titre test is negative. It should be noted that only vaccinations against parvovirus and distemper are considered core vaccines.
First test, then vaccinate! This is the conclusion of the experts and should form the basis of modern vaccination management in dogs. With this strategy, the need for vaccination of each
individual animal can be checked directly in practice.
The antibody titer test thus forms the basis for the establishment of an individual vaccination management and can either be carried out directly in the practice with a rapid test within a few
minutes or a laboratory examination can be requested.
In order to meet the demand of the experts from the vaccination commissions and to build up an individual vaccination management, the demand remains at this point to the pharmaceutical industry,
but also to the licensing authorities, that all 3 vaccines must be available and licensed individually, so that the practitioner can also implement the demand of the WSAVA in practice.
Soon available: Fassisi BoDia + Gia

Diarrhoea diagnostics XXL: antigen detection of rotavirus, coronavirus, E. coli K99, Cryptosporidium parvum and Giardia duodenalis.
Fassisi GiarDia + CPE: The diagnostic duo for more certainty in diarrhoea!

Detection of Giardia duodenalis and Clostridium perfringens enterotoxin (CPE)
Possible causes
Diarrhoea; a frequently seen problem in the veterinary practice. Giardia duodenalis is often the cause. If the intestinal flora becomes unbalanced, physiologically occurring bacteria or viruses
in the intestine can multiply unchecked. These include clostridia, among others.
Their toxin formation can massively worsen the condition of the sick animal. In order to detect this secondary infection at an early stage and to be able to act quickly, Fassisi has developed the
new GiarDia+CPE.
Diagnostic solution
With the new diagnostic duo you can test for Giardia cysts and Clostridium perfringens enterotoxins in parallel. After 10 minutes you know for sure whether one of the two pathogens has been
detected. By using the Fassisi GiarDia+CPE hygienically and at an early stage, targeted measures can be taken directly to quickly alleviate the diarrhoea of the animal and the animal owner.
What to do?
Since Giardia can be transmitted not only from animal to animal, but also from animal to human, quick and consistent action is necessary.
In order to effectively and sustainably combat giardia, it is necessary to interrupt the multiplication cycle through targeted hygiene measures in addition to medicinal treatment.
If the number of intestinal bacteria also increases, this poses a further danger. Clostridia, for example, make the fight against Giardia much more difficult due to their toxin formation.
If CPE is detected in the animal, an attempt can be made to reduce the pathogen to a physiologically occurring level by changing the feed.
The targeted change of feed to combat C. perfringens generally has a very positive effect on the intestinal flora. This is strengthened and built up. As a result, many of the diarrhoea pathogens
(including Giardia) are no longer able to multiply unchecked.
Fassisi CantiCheck Plus: Vaccine titer control made easy

Determination of parvovirus (CPV), canine distemper virus (CDV) and adenovirus (CAV) antibodies in 10 minutes
Vaccinations in dogs
Dogs can be protected from life-threatening viral and bacterial diseases by vaccination. It is important to ensure that vaccine protection is available at all times. At the same time, however,
vaccination should not be given more than necessary in order to administer as few vaccines as possible.
In line with the statement of leading professional associations and vaccination commissions (StIKo Vet) "Vaccinate more animals, the individual animal as often as necessary!", antibody tests
provide an ideal control to prove the duration and status of immunity after vaccination with core vaccines.
The Fassisi CantiCheck Plus
With the Fassisi CantiCheck Plus, an individual vaccination management can be set up to vaccinate the individual animal
only as often as necessary. Only one blood sample from the dog is needed, which can then be checked within a few minutes for the presence of circulating antibodies against canine parvovirus
(CPV), canine distemper virus (CDV) and canine adenovirus (CAV).
For these three parameters, a clear correlation between neutralising antibody levels and protection against clinical disease has been demonstrated.
CantiCheck Plus - Only vaccinate when needed
“Vaccinate as many dogs as possible but only as often as needed”
“Vaccinate as many dogs as possible but only as often as needed” - that’s the motto of leading veterinary associations such as the World Small Animal Veterinary Association (WSAVA). Vaccines continue to be life-savers for people and animals.
Vaccinating as many dogs as possible is essential to reach the so-called herd immunity: in order to avoid epidemics, we should aim for the highest possible vaccination coverage of the dog
population (at least 70%). While this can be achieved by awareness campaigns, the “only vaccinate when needed” part is just as important. And here’s where in-house antibody tests such as the
Fassisi
CantiCheck Plus kit come in.
Tailor-made vaccination protocols
According to the WSAVA vaccination guidelines group, the ‘necessary’ or so-called core vaccines for dogs include those against canine distemper virus (CDV), canine adenovirus (CAV types 1 and 2)
and canine parvovirus (CPV)1. Tailor-made vaccination protocols are key: together with the dog owner, the vet should set up a tailor-made vaccination protocol. What is the age, origin and
lifestyle of the pet? Does it travel or will it spend time in boarding kennels? Is it used for breeding, showing or competitions? Whatever their lifestyle, all dogs will require core vaccines,
defined by the WSAVA as those “that all dogs throughout the world should receive to protect against infectious diseases of global significance”. Additionally, non-core vaccines can be given,
according to the lifestyle.
Core vaccines: not more than once every 3 years
But even core vaccines should not be given needlessly. According to the WSAVA, they should not be given any more frequently than every three years following the primary vaccination series, which
includes the first booster following the puppy series. The reason is simple: the duration of immunity (DOI) is many years and may be up to the lifetime of the pet.
For the core vaccines, the WSAVA guidelines specify that the presence of antibodies – regardless of the titre – indicates protective immunity and immunological memory. They even stress that
“giving more frequent vaccines to animals in an attempt to increase antibody titres is a pointless exercise”.
In-house antibody testing: when to use?
Checking for the presence of antibody for core vaccine components (CDV, CAV, CPV) is therefore a useful tool to determine immune protection, and the use of in-house tests is supported by the
WSAVA guidelines. Ideal tests are fast – providing the result in minutes – and provide a simple “yes/no” answer, such as the Fassisi CantiCheck
Plus kit, which tests for the presence of CDV. CAV and CPV antibodies.
This test has several applications:
- to check the protective immunity in puppies
- to determine the immune status of dogs
- when managing infectious disease outbreaks in shelters
- in case of adverse events
Is my puppy safe? Measuring vaccine take-up
According to the WSAVA guidelines, the third dose of core vaccines should be administered at 16 weeks of age or older. Testing puppies at 20 weeks of age will show which ones are seropositive –
and therefore protected. Seropositive puppies would not require their 26 or 52-week booster and could next receive their core booster 3 years later.
Seronegative puppies should be revaccinated and retested to check they are able to developing protective immunity. Those that fail to respond after revaccination may be either ‘low responders’ or
genetically-determined ‘non-responders’ incapable of mounting an immune response to that antigenic component of the vaccine. Such animals are rare, but certain breeds such as the Rottweiler are
predisposed to developing a less effective immune responses to CPV and rabies virus.
Fassisi has 3 new products in its portfolio!

Fassisi CantiCheck Plus - the Fassisi CanTi Check extended by the detection of antibodies against the canine adenovirus.

Fassisi BoDia + Gia - the Fassisi BoDia extended by the detection of Giardia antigens in cattle.

Another addition to the Fassisi BoDia - the Fassisi BoDia Plus expanded to include detection of Clostridium perfringens antigens in cattle.
Meet us at the 11th Leipzig Veterinary Congress!

07-09/07/2022
Stand: Hall 2 G14
Live Meetings at Trade Shows in November
Meet us at the following Trade Shows:
London Vet Show, 11-12/11/2021
Medica Düssseldorf, 15-18/11/2021
Vet-Congress Berlin, 19-20/11/2021
Study published in „Viruses 2021“: CanTiCheck – Test of Choice
The independent study evaluated the quality and practicability of four point-of-care tests for the detection of anti-CPV antibodies. Specificity was the most significant indicator since the detection is used to show the specific immune status of an adult dog. If antibodies are present, unnecessary vaccinations can be avoided. A total of 241 dog sera were tested. Virus neutralization was used as the reference method.
Due to its specificity of 98% and ease of performance, the CanTiCheck test became the test of choice.
Link to the study
Link
to the article in „Tierärztliche Praxis“
Fassisi has moved
Fassisi has moved to new premises!
Our new home is now at Marie-Curie-Strasse 8, 37079 Goettingen, Germany.
ISO9001:2015

We are proud to annouce that Fassisi is now also certified according to ISO9001:2015.
New test: CanVecto 4

Test for the detection of Dirofilaria immitis antigens, Anaplasma phagocytophilum, antibodies, Ehrlichia canis antibodies and Leishmania antibodies in dogs.
New test: CanVecto 3

Test to detect Anaplasma phagocytophilum, antibodies, Borrelia burgdorferi antibodies
and Erlichia canis antibodies in dogs.
News section under revision.
News section under revision.
8. Leipziger Tierärzte Kongress Leipzig 2016
We would like to thank everyone who visited our stand for the interesting discussions.
It was a pleasure meeting you!

Welcome to our new website
Our website now has a new design (but it is a work in progress).
Fassisi FeLFIV - New Version 2015
The new Version 2015 of our Fassisi FeLFIV is now available. Now with optimized flow characteristics.
The new BoTick is now available
The new revised version of our Fassisi BoTick is now available for the new tick season with better to read test line intensities and an increased sensitivity to detect even lower amounts of borrelia in the tick. The new revised version of our Fassisi BoTick is now available for the new tick season with better to read test line intensities and an increased sensitivity to detect even lower amounts of borrelia in the tick.

New Fassisi FeLFIV with Fassisi-tag
The rapid test for the detection of FIV antibodies and FeLV antigens in cats
Now with the Fassisi-tag for highest test performance and the new pipette for a more accurate test protocol.
For further information, please see Fassisi FeLFIV.

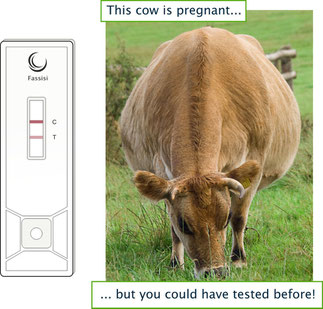
The Fassisi BoviPreg is available
The bovine pregnancy test for the detection of bovine PAG is now available. For more information please see Fassisi BoviPreg.
Fassisi is a certified training company
Fassisi offers vocational training schemes which are audited and certified by the German Chamber of Commerce & Industry (IHK).
Pre-announcement: The Fassisi tag comes
We are working on innovations for your on-site diagnostics. We optimize our tests to meet your requirements. Soon, our antibody tests can be applied in the practice much broader due to the use of the patented Strep-Tag technology.

Fassisi BoTick Version 2013 now available
The testing of ticks for Borrelia is now even easier. Due to the new test setup the sensitivity of the Fassisi BoTick of the test could be increased further.
Also thanks to the new test cassette weak test lines can be better recognized and the tick sample fluid runs more harmoniously over the test strip.
Commission work
Fassisi now develops and manufactures customer-specific products on a commission basis using our innovative technologies.
Contact us for more information.
The new Fassisi Giardia latex is now available
The new Fassisi GiarDia with blue latex as detection particals is now availible.
Due to a new coupling technique based on latex particles, the test results are available in a few minutes in a deep blue, which makes the test easier and more accurate to read. Even low concentrations of Giardia cysts are clearly recognized, which enables a fast and reliable detection of Giardia duodenalis (lamblia) antigens in feces of dogs and cats.
Please see Fassisi GiarDia for further information.
Fassisi BoTick detects B. spielmanii
The Fassisi BoTick also detects the borrelia species Borrelia spielmanii. Thus, the Fassisi BoTick is the only rapid test, which detects the species B. burgdorferi, B. garinii, B. afzelii and B. spielmanii directly in the tick.

Products registered by the FLI
Fassisi has registered three more products in Germany to sell in the German market with the FLI:
- Fassisi FeLV - as single test derived from the Fassisi FeLFIV double test
- Fassisi PiDia Light - as test for the use for large animals
- Fassisi LymeBo - for the use in dogs
New product: Fassisi Triple
The Fassisi Triple Test for dogs and cats is now available. Fassisi Triple, the rapid test for the reliable and easy on-site detection of Parvovirus (FPV, CPV), Corona virus (FCoV, CCV) and Giardia in feces of dogs or cats.
Please contact us for further information.
Fassisi receives the GMP Certificate
The Fassisi GmbH has received an extension of the manufacturer authorization according to § 13 AMG (German Pharmaceuticals Act) and is now entitled to manufacture and distribute a wider range of medicinal products according to the German Pharmaceuticals Act.
Furthermore, the Fassisi GmbH has received the certification according to GMP guidelines (good manufacturing practice) .
New product: Bovine allergens
The rapid test AllerBovine is for the on-site detection of elevated concentrations of bovine allergens in homes from dust samples. The test supports i.a. preventive measures to reduce by bovine allergens caused respiratory diseases. The test was developed in cooperation with the University of Goettingen.
Further information will follow later.
New products from Fassisi
New product developments:
Livestock
Fassisi IgG calf - the rapid test for the semi-quantitative determination of the IgG level in new born calves in the ranges 0-5, 5-10 and >10 mg/ml in serum
Fassisi BoDia light - the rapid test for calf scours for the parallel detection of Clostridium perfringens and Rotavirus in faeces of calves
Companion animals
Fassisi EhrCanis - the rapid test for the detection Ehrlichia canis antibodies in whole blood, serum, plasma of dogs
Fassisi LymeBo - the rapid test for the detection of Borrelia burgdorferi antibodies in whole blood, serum, plasma of dogs
Now availible as single tests:
Companion animals
Fassisi FeLV - the rapid test for the detection of FeLV antigens in whole blood, serum, plasma of cats
Fassisi FIV - the rapid test for the detection of FIV antibodies in whole blood, serum, plasma of cats
Fassisi Parvo - the rapid test for the detection of Parvovirus in faeces of dogs and cats
Fassisi Corona - the rapid test for the detection of Coronavirus in faeces of dogs and cats
New product combination:
Fassisi Triple - the rapid test for the parallel detection of Parvovirus, Coronavirus and Giardia in faecal samples of dogs and cats
Further information will follow soon.
Fassisi is granted a manufacturing licence in accordance with the German Medicinal Products Act
Fassisi GmbH has been granted a manufacturing licence in accordance with section 13 of the German Medicinal Products Act (Arzneimittelgesetz – AMG) and is therefore now authorised to manufacture and distribute compliant medicinal products.
These manufacturing licences are necessary for the production of rapid veterinary test systems in Germany and also safeguard the high quality standards associated with the “Made in Germany” label.
The Fassisi TetaCheck is now availible
The Fassisi TetaCheck, the rapid test est to check the tetanus
antibody titer in horses is now available.

Fassisi wins the Bio-founder competition 2011
On 27 September 2011 in Boenen, North Rhein-Westphalia, Fassisi was awarded first place in the Bio-founder Competition 2011 for an innovative rapid test product and the corresponding marketing concept.
We would like to say thank you for this award and look forward to the imminent market launch of Fassisi Tetacheck.
Welcome on our new homepage
We are looking forward to your comments and suggestions.
Contact
Fassisi, Gesellschaft für Veterinärdiagnostik und Umweltanalysen mbH
Marie-Curie-Strasse 8
37079 Göttingen
Germany
info@fassisi.de
Phone: +49 (0)551 5008840





